Did a 2-Year-Old Die After Receiving a Dose of the Pfizer Vaccine?
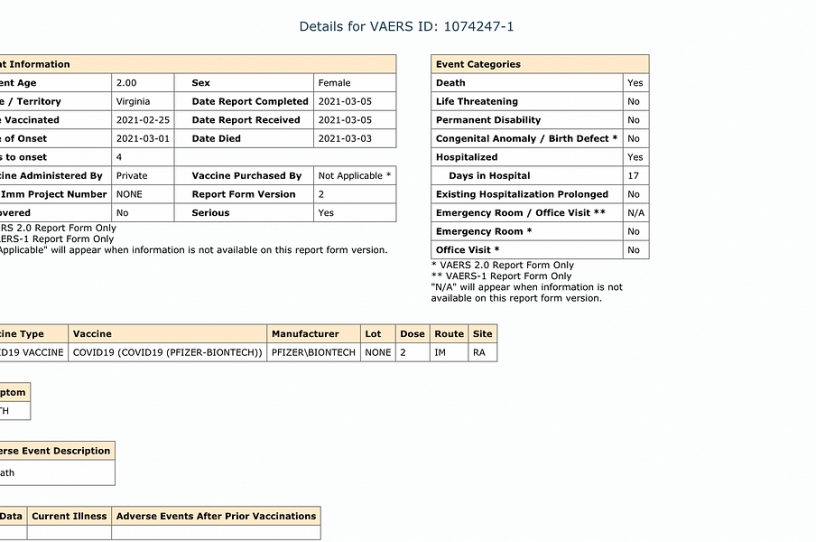
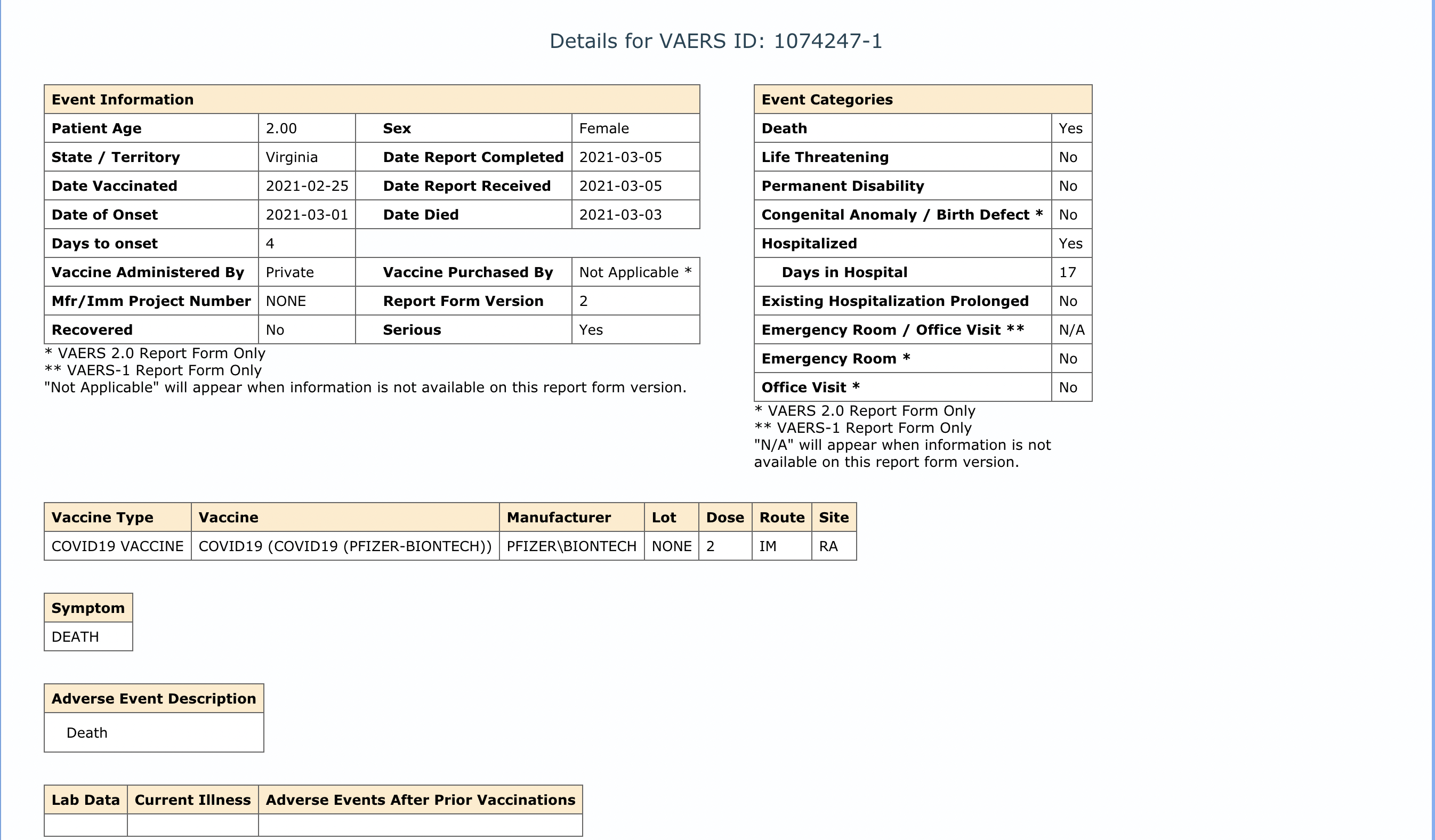
Viral social media posts and articles claim that a 2-year-old passed away after receiving the Pfizer coronavirus vaccine. This claim relies on an entry in the U.S. Vaccine Adverse Events Reporting System (VAERS), which details the alleged death of a 2-year-old girl in Virginia who received the Pfizer coronavirus vaccine on February 25 and died away a few days later on March 3 after being hospitalized for 17 days.
VAERS is a system managed by the Centers for Disease Control and Prevention and the Food and Drug Administration through which individuals may submit reports of side effects after receiving a vaccine. As noted on the VAERS website, anyone is able to submit a report to VAERS; “VAERS is a passive reporting system, meaning it relies on individuals to send in reports of their experiences to CDC and FDA. VAERS is not designed to determine if a vaccine caused a health problem, but is especially useful for detecting unusual or unexpected patterns of adverse event reporting that might indicate a possible safety problem with a vaccine.”
In other words, the reports filed through VAERS are not indicative of actual vaccine side effects, they’re simply used as a jumping off point for investigating possible adverse effects. It may turn out that a previously unknown adverse effect is tied to the vaccine in question, though the reported adverse event may turn out to be entirely unrelated to the vaccine. It’s also entirely possible for false reports to be filed with VAERS—Vice shared the story of anesthesiologist James Laidler, who filed a VAERS report claiming a flu shot turned him into the Hulk. Laidler did so to show why unverified reports in VAERS should be taken with a grain of salt.
Beyond the reasons to be skeptical of any VAERS report, the story of the 2-year-old girl dying after receiving the Pfizer vaccine contains some obvious red flags, namely that Pfizer hadn’t even begun clinical trials with children under the age of 11 at the time. It wasn’t until March 2021 that Pfizer started trials with children ages 6 months to 11 years. Moderna began its clinical trials with young children in March as well. Since these trials have not been completed, the Pfizer vaccine has not been authorized for anyone under the age of 16. The report also states that the child spent 17 days in the hospital; given that she allegedly received the vaccine on February 25 and passed away six days later, the bulk of this hospitalization would have occurred before the vaccine was even allegedly administered.
VAERS reports should be viewed skeptically in general—even if reports are accurate they aren’t necessarily indicative of vaccines causing adverse events. And no 2-year-old in the United States would have received the Pfizer vaccine in February, leaving little reason to believe that the VAERS report behind these viral reports is accurate.
If you have a claim you would like to see us fact check, please send us an email at factcheck@thedispatch.com. If you would like to suggest a correction to this piece or any other Dispatch article, please email corrections@thedispatch.com.